This is a titration method where a measured amount of sodium hydroxide is added to the test sample until the end point is reached. The end point can be determined two ways; by reaching a pH of 8.2 using a pH meter or by use of an indicator (phenolphthalein) that changes to a magenta color once the pH reaches 8.2. The ASBC method describes both of these options.

Figure 1 - Click to Enlarge
A titration is a lab method where a solution of a known concentration (the titrant) is used to determine the concentration of an unknown solution (the titrand). In this method the titrant is 0.1M sodium hydroxide and the titrand is the beer sample. The set up looks similar to what is shown in Figure 1.
This method describes a weak acid – strong base titration. This is where a strong base (sodium hydroxide) is added to a solution containing a weak acid (beer sample) until all of the acid’s hydrogens have been neutralized. Neutralization is the quantitative process where the hydrogen ions (H+) of the acid and the hydroxide ions (OH-) of the base react to form water and a salt until no more acid is left in solution.
acid + base → salt + water
HCl + NaOH → NaCl + H2O
The pH of when this point occurs, known at the equivalence point, depends on the strength of the acid within the reaction and is not always a neutral pH of 7.
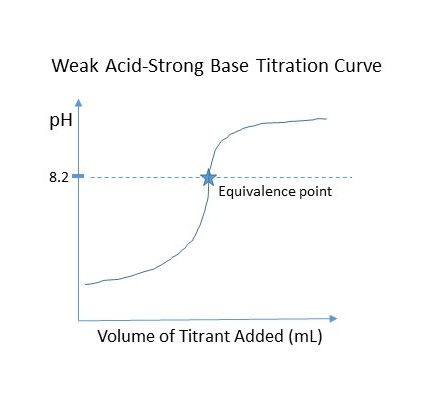
Figure 2 - Click to Enlarge
The equivalence point is also called the end point of the titration. In a weak acid-strong base titration the equivalence point is greater than pH 7. In this method the equivalence point is reached at 8.2 pH. A titration curve plots the pH vs. the volume of the titrant added (Figure 2), where the equivalence point is the middle vertical spot on the curve. Notice how the rate of pH increase quickens the closer it gets to the equivalence point. In practice this means the pH will increase faster the closer to the end point and is the reason why the method describes adding smaller volumes once pH 7.6 is reached. This can’t be determined if using the indicator method.
There are many indicators available for titrations but for this method phenolphthalein is the best choice if a pH meter cannot be used because it changes color and appears magenta under basic conditions in the range of 8.2 -10 pH, which is this method’s equivalence point. Once the titration has reached its equivalence a small excess amount of the base will create a basic environment in which the indicator is will appear magenta, signaling the end point of the titration.
Organic acids that lead to flavor characteristics in beer (for example: lactic, acetic, malic, and citric acid) are considered weak acids and they do not dissociate completely. This means that not all of the hydrogens/protons on the molecule will unbind and react with the base that is working to neutralize the solution in a titration.
HA ↔ A- + H+
During the titration process, all of the acidic hydrogens from all organic acids in the solution are reacting with the base resulting in titratable acidity. Although this value can be represented in units of a specific organic acid (for example: g/L or percent % lactic acid), it does not mean that all of the acids in solution are lactic acid. This is misleading but it is one way to report the results.
The correction factors come from the molar mass of each organic acid divided by the number of acidic protons, where the acidic protons are the hydrogens in an acid what will dissociate. This is described in Table 1.
|
Lactic | 90 | 1 | 90 |
|---|
|
Acetic | 60 | 1 | 60 |
|---|
|
Malic | 134 | 2 | 67 |
|---|
|
Citric | 192 | 3 | 64 |
|---|
Table 1
Green Chemistry
|
Green Action |
Principle |
Benefit |
Choose to determine TA by pH meter
|
Safer Solvents | Removes the use of Phenolphthalein, a hazardous chemical |

Energy Efficient | Removes the use of a heating mantle/hot plate |

Prevent Waste | Removes the use of distilled water |

Prevent Accidents
| Removes hot glass and hazardous chemicals |
Image credit: iconsmind.com
Big Thanks to...
Ann Sandbrook, Joe Palausky, Dana Garves & Katie Fromuth